Paul DeLeo, PhD, Integral Consulting06.01.18
In the nineteenth century, Hans Christian Andersen told the story of a conceited emperor who was willing to believe that his nonexistent new suit of clothes was visible only to the worthiest of his subjects. It took a child in the crowd to confront the false pretext of the invisible clothing and shatter the emperor’s illusion.
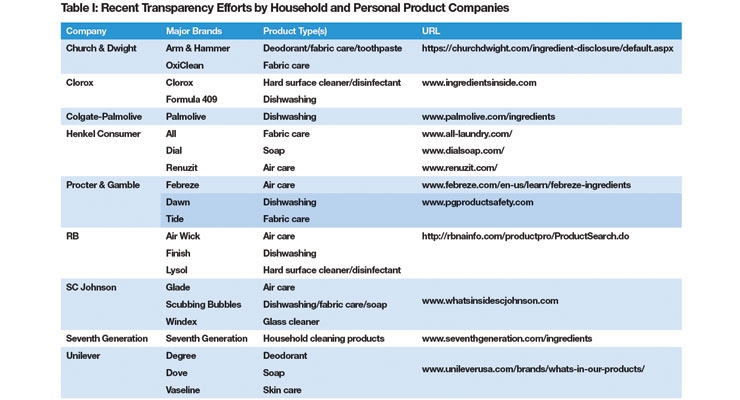
Much the same, manufacturers of household consumer products are being confronted by a public that is no longer entirely willing to go along with brand imagery and is increasingly looking for substance to support the value proposition of any product. Consumers and activists are demanding more information regarding what is in products and how they are made. They have taken those demands to legislators and regulators but are turning more frequently to retailers to force product manufacturers to disclose more. During the summer of 2017, legislators in California and regulators in New York State were in a race to be the first to enact rules requiring the disclosure of ingredients in consumer and institutional cleaning products despite current transparency efforts by leading consumer product manufacturers (see Table 1). California won, by the way, and currently is implementing the Cleaning Product Right to Know Act of 2017.
The recent frenzy over cleaning product ingredient disclosure was a symptom of a phenomenon that has been playing out since the early 2000s as access to the internet exploded and social media blossomed. The term radical transparency has been used to describe actions in politics, government, and business where the boundaries of what may be considered confidential are being tested and eroded, and the means to disseminate information are more accessible. All businesses, whether product or service providers, are being challenged to address changing consumer expectations in an era of increasing transparency, and there is a fundamental question—will one react to those trends, or get ahead of them and take advantage of the business opportunity?
The following cautionary tale underlines how a company can easily lose control of its product. In 2012, Vani Hari, an American blogger calling herself “the Food Babe” identified azodicarbonamide as an ingredient in Subway sandwich bread in North America. That ingredient was also used in the production of yoga mats and other durable materials. Despite limited resources and no apparent training in public health, she was able to steal the safety narrative from a major corporation and, at a minimum, make the lives of some corporate executives very uncomfortable for several weeks. The lesson is that there are few barriers today for anyone creating a narrative about a product, and companies are vulnerable to having the likes of the Food Babe tell their safety story if they have not created and disseminated one themselves.
While a comprehensive and even compelling safety story is no guarantee of continued success in the market, the lack of a substantive safety profile (at your fingertips) is a recipe for defeat in the face of crisis. A recent example is the rapidity with which plastic microbeads were banned in wash-off personal care products, due in some measure to a lack of environmental safety data—either because such data are not required for most cosmetics or personal care products, or because most users of the materials assumed the inert nature of these products rendered development of comprehensive environmental safety information unnecessary.
The Inventory Story
Every consumer product manufacturer will have a comprehensive ingredient portfolio from which it is formulating existing products or developing new ones. Typically, the manufacturer will have an established process for adding new raw materials and ingredients to its portfolio, and the ingredient inventory is a basis from which hazard communication information is developed. The ingredient inventory is the platform for a comprehensive ingredient safety communication program.
It is incumbent upon product manufacturers to understand the vulnerabilities and potential liabilities associated with their ingredient portfolio. There are a number of simple approaches one may use identify those vulnerabilities.
The Chemical Footprint Project (CFP) is a transparency initiative from a group of long-time “safer chemicals” partners that seeks to “transform global chemical use by measuring and disclosing data on business progress to safer chemicals” (www.chemicalfootprint.org). It uses a questionnaire approach to encourage companies to develop an internal chemical management program and to consider potential hazardous chemicals for elimination from their chemical inventory. Regardless of whether you ascribe to the philosophical perspective underpinning the program, there are a number of good questions proposed that can assist in better understanding potential vulnerabilities and liabilities.
At the heart of the CFP survey are fundamental questions regarding materials management:
LoL Is No Laughing Matter
One relatively quick way to screen potential threats is a vulnerability assessment of the ingredient portfolio. A common technique for assessing vulnerability is to compare the ingredients in the portfolio against various hazard lists generated by respected organizations—authoritative bodies such as the International Agency for Research on Cancer, government agencies like the California Office of Environmental Health Hazard Assessment, and scientific and professional bodies such as the Association of Occupational and Environmental Clinics (AOEC). This approach is often referred to as a list of lists (LoL) approach. Many retailers maintain internal LoLs and “encourage” the suppliers of the consumer products they stock to adapt those products by deselecting particular ingredients found on those lists. A regulatory example of the LoL approach is that used by the California Department of Toxic Substances Control (DTSC) in its Safer Consumer Products program to identify potential candidate chemicals of concern. DTSC uses 15 hazard trait lists and 8 exposure potential lists containing more than 4,000 chemicals.
A significant concern with the LoL approach is that many of the organizations developing lists do not have open and transparent processes whereby new or existing data that were previously not publicly available might be brought forward. Moreover, lists that are being developed by professional societies to assist clinicians in the field, such as the Occupational Asthmagens on the AOEC List of Exposure Codes, are being picked up by regulatory authorities for risk management without consideration of the purpose of the list and its appropriateness for regulatory application. These trends do not seem to be abating, and there is no simple solution for makers and users of chemicals beyond vigilance and proactive internal chemical management strategies.
Think Inside the Box, Bottle or Jar
A more proactive approach to understanding vulnerabilities and potential liabilities within a chemical portfolio is to conduct a human and environmental safety assessment for every ingredient within one’s portfolio. While it is common for cosmetic and home care product manufacturers to conduct a human and environmental safety assessment for each ingredient used in each product formulation before the product enters the market, those processes typically may not be designed to withstand external scrutiny and, consequently, may not provide the kind of support needed when a crisis hits. In an era of radical transparency, product manufacturers should consider external communication of chemical safety information as they are conducting those internal assessments.
A recent example of this approach was an effort by the American Cleaning Institute (ACI) to collect and make publicly available complete human and environmental safety information for each ingredient used in consumer cleaning products in the US as part of its Cleaning Product Ingredient Safety Initiative (https://www.cleaninginstitute.org/cpisi/). The intent of the program was not only to collect the information, but to make it publicly available and to provide a risk-based safety profile of the chemical based on its use in the associated cleaning products. Other recent examples include a transparency initiative from the international crop protection industry to make pesticide safety information more readily available, and the SmartLabel initiative developed by product manufacturers and their retail outlets to create web-based and mobile apps that allow consumers to access information on nutrients, ingredients, allergens, and other pertinent aspects of a variety of food, beverage, household and personal care products.
A Range of Screening Tests
A major concern of anyone contemplating the development of a safety profile for each chemical in a portfolio will be the acquisition of the data necessary to assess the likely hundreds of chemicals and possibly dozens of exposure scenarios. Driven by the desire to replace vertebrate animals in safety testing, a number of what are now referred to as new approach methodologies (aka NAMs) are providing tools and publicly available data sources for potential use in safety assessments. In chemico, in vitro, in silico sounds like something a twenty-first century Julius Caesar might say in conquering scientific questions, but it refers to abiotic laboratory screening tests, biologically based screening tests, and computational models and database analysis, respectively, which generate data regarding the safety of chemicals (see box). Several European and US government programs have conducted and released data from in chemico and in vitro laboratory screening assays in recent years. A number of computational tools and databases are now available from US EPA and the European Chemicals Agency (ECHA), including quantitative structure-property relationship (QSPR) models for the generation of physical-chemical and environmental fate data, and quantitative structure-activity relationship (QSAR) models for ecotoxicological and human toxicological data. Perhaps more interesting from ECHA is the release of REACH Summary Data in April 2017, which includes the physical-chemical properties, environmental fate, and human and environmental endpoint summary data for 15,000 chemicals that have been registered under the program. This database allows one to understand the human and environmental safety of the chemicals within the database, but it also facilitates read-across of potentially thousands of additional chemicals.
New tools and the availability of data for thousands of existing chemicals makes it easier than ever for a company to develop a safety narrative for every chemical it uses. The alternative is to wait for someone else to tell that story… but don’t expect it to be favorable.
Keep Calm and…
In the era of radical transparency, it is critical that the chemically intensive enterprise know all of the chemicals present in its chemical portfolio, know the safety information associated with each chemical, create a safety narrative for each chemical, and share that information throughout and beyond the value chain with customers, consumers, regulators, and other stakeholders. At the end of the day, a cloak of transparency may be the best cover.
Dr. Paul DeLeo is a scientific professional whose focus over the past two decades has included applied research, regulatory advocacy and public outreach related to the human and environmental safety of food ingredients, pesticides, pharmaceutical and personal care products, and cleaning products. A strategic and visionary thinker, Dr. DeLeo successfully implements solutions from conception through rollout and broad stakeholder outreach. Dr. DeLeo currently serves as a Principal with Integral Consulting Inc. in the Washington, D.C. metro area where he leads the firm’s Product Stewardship practice.
More info: Paul C. DeLeo, Ph.D., Integral Consulting Inc., 200 Harry S. Truman Parkway, Suite 330, Annapolis, MD 21401, Tel: 410.573.1982, ext. 515; Email: pdeleo@integral-corp.com;
Website: www.integral-corp.com
Much the same, manufacturers of household consumer products are being confronted by a public that is no longer entirely willing to go along with brand imagery and is increasingly looking for substance to support the value proposition of any product. Consumers and activists are demanding more information regarding what is in products and how they are made. They have taken those demands to legislators and regulators but are turning more frequently to retailers to force product manufacturers to disclose more. During the summer of 2017, legislators in California and regulators in New York State were in a race to be the first to enact rules requiring the disclosure of ingredients in consumer and institutional cleaning products despite current transparency efforts by leading consumer product manufacturers (see Table 1). California won, by the way, and currently is implementing the Cleaning Product Right to Know Act of 2017.
The recent frenzy over cleaning product ingredient disclosure was a symptom of a phenomenon that has been playing out since the early 2000s as access to the internet exploded and social media blossomed. The term radical transparency has been used to describe actions in politics, government, and business where the boundaries of what may be considered confidential are being tested and eroded, and the means to disseminate information are more accessible. All businesses, whether product or service providers, are being challenged to address changing consumer expectations in an era of increasing transparency, and there is a fundamental question—will one react to those trends, or get ahead of them and take advantage of the business opportunity?
The following cautionary tale underlines how a company can easily lose control of its product. In 2012, Vani Hari, an American blogger calling herself “the Food Babe” identified azodicarbonamide as an ingredient in Subway sandwich bread in North America. That ingredient was also used in the production of yoga mats and other durable materials. Despite limited resources and no apparent training in public health, she was able to steal the safety narrative from a major corporation and, at a minimum, make the lives of some corporate executives very uncomfortable for several weeks. The lesson is that there are few barriers today for anyone creating a narrative about a product, and companies are vulnerable to having the likes of the Food Babe tell their safety story if they have not created and disseminated one themselves.
While a comprehensive and even compelling safety story is no guarantee of continued success in the market, the lack of a substantive safety profile (at your fingertips) is a recipe for defeat in the face of crisis. A recent example is the rapidity with which plastic microbeads were banned in wash-off personal care products, due in some measure to a lack of environmental safety data—either because such data are not required for most cosmetics or personal care products, or because most users of the materials assumed the inert nature of these products rendered development of comprehensive environmental safety information unnecessary.
The Inventory Story
Every consumer product manufacturer will have a comprehensive ingredient portfolio from which it is formulating existing products or developing new ones. Typically, the manufacturer will have an established process for adding new raw materials and ingredients to its portfolio, and the ingredient inventory is a basis from which hazard communication information is developed. The ingredient inventory is the platform for a comprehensive ingredient safety communication program.
It is incumbent upon product manufacturers to understand the vulnerabilities and potential liabilities associated with their ingredient portfolio. There are a number of simple approaches one may use identify those vulnerabilities.
The Chemical Footprint Project (CFP) is a transparency initiative from a group of long-time “safer chemicals” partners that seeks to “transform global chemical use by measuring and disclosing data on business progress to safer chemicals” (www.chemicalfootprint.org). It uses a questionnaire approach to encourage companies to develop an internal chemical management program and to consider potential hazardous chemicals for elimination from their chemical inventory. Regardless of whether you ascribe to the philosophical perspective underpinning the program, there are a number of good questions proposed that can assist in better understanding potential vulnerabilities and liabilities.
At the heart of the CFP survey are fundamental questions regarding materials management:
- Do you know what chemicals you use?
- Do you understand the chemicals that you use?
- How would you handle public scrutiny of the chemicals you use (from academics, regulators, downstream customers, consumers)?
LoL Is No Laughing Matter
One relatively quick way to screen potential threats is a vulnerability assessment of the ingredient portfolio. A common technique for assessing vulnerability is to compare the ingredients in the portfolio against various hazard lists generated by respected organizations—authoritative bodies such as the International Agency for Research on Cancer, government agencies like the California Office of Environmental Health Hazard Assessment, and scientific and professional bodies such as the Association of Occupational and Environmental Clinics (AOEC). This approach is often referred to as a list of lists (LoL) approach. Many retailers maintain internal LoLs and “encourage” the suppliers of the consumer products they stock to adapt those products by deselecting particular ingredients found on those lists. A regulatory example of the LoL approach is that used by the California Department of Toxic Substances Control (DTSC) in its Safer Consumer Products program to identify potential candidate chemicals of concern. DTSC uses 15 hazard trait lists and 8 exposure potential lists containing more than 4,000 chemicals.
A significant concern with the LoL approach is that many of the organizations developing lists do not have open and transparent processes whereby new or existing data that were previously not publicly available might be brought forward. Moreover, lists that are being developed by professional societies to assist clinicians in the field, such as the Occupational Asthmagens on the AOEC List of Exposure Codes, are being picked up by regulatory authorities for risk management without consideration of the purpose of the list and its appropriateness for regulatory application. These trends do not seem to be abating, and there is no simple solution for makers and users of chemicals beyond vigilance and proactive internal chemical management strategies.
Think Inside the Box, Bottle or Jar
A more proactive approach to understanding vulnerabilities and potential liabilities within a chemical portfolio is to conduct a human and environmental safety assessment for every ingredient within one’s portfolio. While it is common for cosmetic and home care product manufacturers to conduct a human and environmental safety assessment for each ingredient used in each product formulation before the product enters the market, those processes typically may not be designed to withstand external scrutiny and, consequently, may not provide the kind of support needed when a crisis hits. In an era of radical transparency, product manufacturers should consider external communication of chemical safety information as they are conducting those internal assessments.
A recent example of this approach was an effort by the American Cleaning Institute (ACI) to collect and make publicly available complete human and environmental safety information for each ingredient used in consumer cleaning products in the US as part of its Cleaning Product Ingredient Safety Initiative (https://www.cleaninginstitute.org/cpisi/). The intent of the program was not only to collect the information, but to make it publicly available and to provide a risk-based safety profile of the chemical based on its use in the associated cleaning products. Other recent examples include a transparency initiative from the international crop protection industry to make pesticide safety information more readily available, and the SmartLabel initiative developed by product manufacturers and their retail outlets to create web-based and mobile apps that allow consumers to access information on nutrients, ingredients, allergens, and other pertinent aspects of a variety of food, beverage, household and personal care products.
A Range of Screening Tests
A major concern of anyone contemplating the development of a safety profile for each chemical in a portfolio will be the acquisition of the data necessary to assess the likely hundreds of chemicals and possibly dozens of exposure scenarios. Driven by the desire to replace vertebrate animals in safety testing, a number of what are now referred to as new approach methodologies (aka NAMs) are providing tools and publicly available data sources for potential use in safety assessments. In chemico, in vitro, in silico sounds like something a twenty-first century Julius Caesar might say in conquering scientific questions, but it refers to abiotic laboratory screening tests, biologically based screening tests, and computational models and database analysis, respectively, which generate data regarding the safety of chemicals (see box). Several European and US government programs have conducted and released data from in chemico and in vitro laboratory screening assays in recent years. A number of computational tools and databases are now available from US EPA and the European Chemicals Agency (ECHA), including quantitative structure-property relationship (QSPR) models for the generation of physical-chemical and environmental fate data, and quantitative structure-activity relationship (QSAR) models for ecotoxicological and human toxicological data. Perhaps more interesting from ECHA is the release of REACH Summary Data in April 2017, which includes the physical-chemical properties, environmental fate, and human and environmental endpoint summary data for 15,000 chemicals that have been registered under the program. This database allows one to understand the human and environmental safety of the chemicals within the database, but it also facilitates read-across of potentially thousands of additional chemicals.
New tools and the availability of data for thousands of existing chemicals makes it easier than ever for a company to develop a safety narrative for every chemical it uses. The alternative is to wait for someone else to tell that story… but don’t expect it to be favorable.
Keep Calm and…
In the era of radical transparency, it is critical that the chemically intensive enterprise know all of the chemicals present in its chemical portfolio, know the safety information associated with each chemical, create a safety narrative for each chemical, and share that information throughout and beyond the value chain with customers, consumers, regulators, and other stakeholders. At the end of the day, a cloak of transparency may be the best cover.
Dr. Paul DeLeo is a scientific professional whose focus over the past two decades has included applied research, regulatory advocacy and public outreach related to the human and environmental safety of food ingredients, pesticides, pharmaceutical and personal care products, and cleaning products. A strategic and visionary thinker, Dr. DeLeo successfully implements solutions from conception through rollout and broad stakeholder outreach. Dr. DeLeo currently serves as a Principal with Integral Consulting Inc. in the Washington, D.C. metro area where he leads the firm’s Product Stewardship practice.
More info: Paul C. DeLeo, Ph.D., Integral Consulting Inc., 200 Harry S. Truman Parkway, Suite 330, Annapolis, MD 21401, Tel: 410.573.1982, ext. 515; Email: pdeleo@integral-corp.com;
Website: www.integral-corp.com